Articles
Article Tools
Supplementary
Stats or Metrics
Article
Original Article
Exp Neurobiol 2019; 28(4): 529-536
Published online August 31, 2019
https://doi.org/10.5607/en.2019.28.4.529
© The Korean Society for Brain and Neural Sciences
Calpain-2 as a Treatment Target in Prenatal Stress-induced Epileptic Spasms in Infant Rats
Hyeok Hee Kwon1,2,4, Chiranjivi Neupane1,5, Juhee Shin1,2, Do Hyeong Gwon1,2, Yuhua Yin1,2, Nara Shin1,2, Hyo Jung Shin1,2,3, Jinpyo Hong2,3, Jin Bong Park1,3,5, YoonYoung Yi4, Dong Woon Kim1,2,3* and Joon Won Kang1,3,4*
1Department of Medical Science, Chungnam National University, Daejeon 35015, 2Department of Anatomy, School of Medicine, Chungnam National University, Daejeon 35015, 3Brain Research Institute, School of Medicine, Chungnam National University, Daejeon 35015, 4Department of Pediatrics, Chungnam National University Hospital, Daejeon 35015, 5Department of Physiology, School of Medicine, Chungnam National University, Daejeon 35015, Korea
Correspondence to: *To whom correspondence should be addressed.
Dong Woon Kim, TEL: 82-42-580-8209, FAX: 82-42-586-4800
e-mail: visnu528@cnu.ac.kr
Joon Won Kang, TEL: 82-42-280-8244, FAX: 82-42-255-3158
e-mail: childlove@cnu.ac.kr
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License(http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, andreproduction in any medium, provided the original work is properly cited.
Abstract
Stress can induce a serious epileptic encephalopathy that occurs during early infancy. Recent studies have revealed that prenatal stress exposure is a risk factor for the development of infantile spasms. Our previous work demonstrates that prenatal stress with betamethasone-induced alterations to the expression of the K+/Cl− co-transporter (KCC2) in gamma-aminobutyric acid (GABA) interneurons lowers the seizure threshold in exposed animals. Here, we further investigated the mechanisms involved in this KCC2 dysfunction and explored possible treatment options. We stressed Sprague-Dawley rats prenatally and further treated dams with betamethasone on gestational day 15, which increases seizure susceptibility and NMDA (N-Methyl-D-aspartate)-triggered spasms on postnatal day 15. In this animal model, first, we evaluated baseline calpain activity. Second, we examined the cleavage and dephosphorylation of KCC2. Finally, we checked the effect of a calpain inhibitor on seizure occurrence. The phosphorylated-N-methyl-D-aspartate Receptor 2B (NR2B):non-phosphorylated NR2B ratio was found to be higher in the cortex of the prenatally stressed beta-methasone model. We further found that the betamethasone model exhibited increased phosphorylation of calpain-2 and decreased phosphorylation of KCC2 and Glutamic acid decarboxylase 67 (GAD67). After using a calpain inhibitor in prenatal-stress rats, the seizure frequency decreased, while latency increased. GABAergic depolarization was further normalized in prenatal-stress rats treated with the calpain inhibitor. Our study suggests that calpain-dependent cleavage and dephosphorylation of KCC2 decreased the seizure threshold of rats under prenatal stress. Calpain-2 functions might, thus, be targeted in the future for the development of treatments for epileptic spasms.
Graphical Abstract
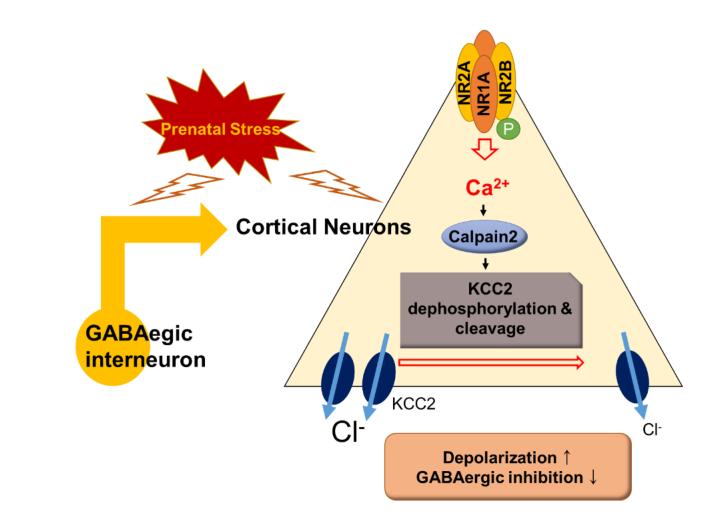
Keywords: Epilepsy, Calpain, KCC2, NMDA, Glutamate decarboxylase 67, K+/Cl- co-transporter


