Articles
Article Tools
Supplementary
Stats or Metrics
Article
Original Article
Exp Neurobiol 2019; 28(6): 697-708
Published online December 31, 2019
https://doi.org/10.5607/en.2019.28.6.697
© The Korean Society for Brain and Neural Sciences
Recapitulation of Neuropsychiatric Behavioral Features in Mice Using Acute Low-dose MK-801 Administration
Darine Froy N. Mabunga1, Donghyun Park1, Onjeon Ryu1, Schley T. Valencia1, Keremkleroo Jym L. Adil1, Seonmin Kim1, Kyoung Ja Kwon1, Chan Young Shin1,2* and Se Jin Jeon1*
1Department of Neuroscience, School of Medicine, Konkuk University, 2Department of Pharmacology, School of Medicine, Konkuk University, Seoul 05029, Korea
Correspondence to: *To whom correspondence should be addressed.
Chan Young Shin, TEL: 82-2-2030-7834, FAX: 82-2-2030-7899
e-mail: chanyshin@kku.ac.kr
Se Jin Jeon, TEL: 82-2-454-5630, FAX: 82-2-2030-7899
e-mail: sjinjeon@hanmail.net
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License
(http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and
reproduction in any medium, provided the original work is properly cited.
Abstract
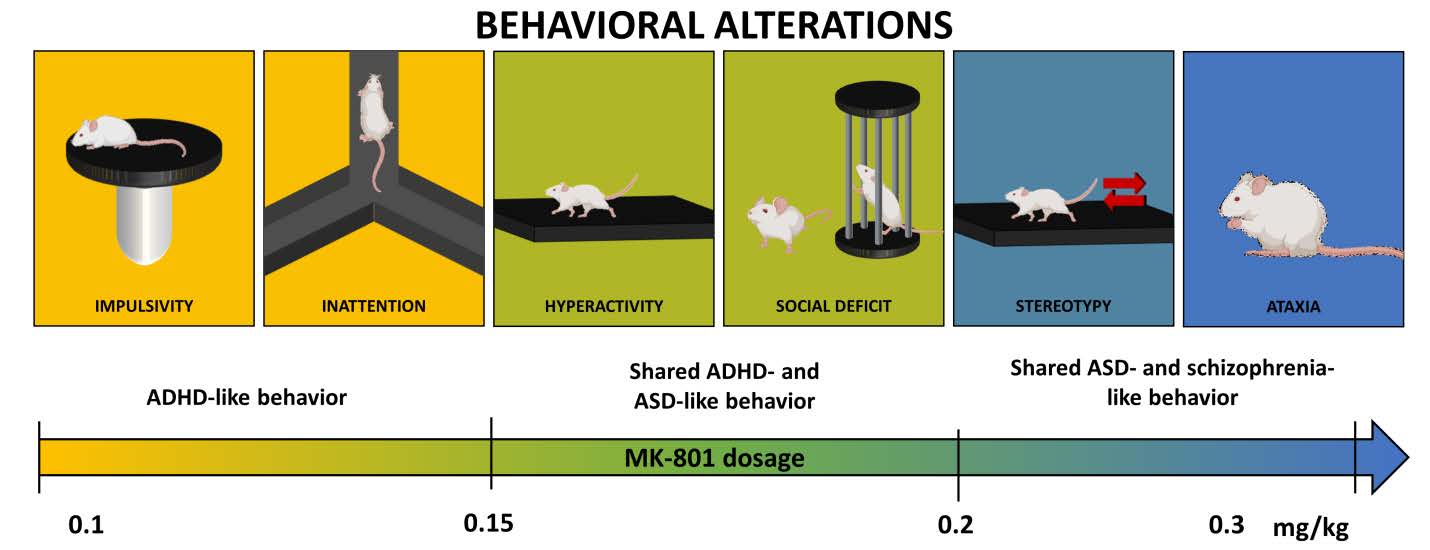
Despite some innate limitations, animal models are a potent investigative tool when used to model specific symptoms of a disorder. For example, MK-801, an N-methyl-D-aspartate receptor antagonist, is used as a pharmacological tool to induce symptoms found in some neuropsychiatric disorders. However, a close examination of literature suggests that the application window of MK-801 doses is relatively narrow between individual behavioral paradigms, necessitating careful characterization of the evoked behavioral aberrations and the doses used to induce them. Moreover, variation in behaviors depending on the animal strain, gender of the subject, and the timing of administration is observed, making it difficult to compare the behavioral characteristics reported in different studies. We aim to characterize the behavioral aberrations induced by different doses of MK-801 in CD-1 mice and create a ready reference for future studies. We used CD-1 mice to recapitulate behavioral impairments resulting from acute administration of MK-801. In 0.1 mg kg−1, we observed diminished spontaneous alteration during the Y-maze test, while 0.12 mg kg−1 resulted in hyperlocomotion and social deficit. Mice treated with 0.2 and 0.3 mg kg−1 of MK-801 demonstrated a decreased self-grooming. Finally, all doses significantly impaired cliff avoidance behaviors suggesting increased impulsivity. These results affirm that MK-801 can effectively model various symptoms of different neuropsychiatric disorders in a dose-dependent manner. The observed sensitivity against spatial-memory impairment and impulsive behaviors at low concentration of MK-801 suggest that MK801 may modulate cognitive function and impulsivity in even lower concentration before it can modulate other behavioral domains.
Graphical Abstract
Keywords: MK-801, Behavioral domains, Neuropsychiatric disorders, Animal model


