Articles
Article Tools
Stats or Metrics
Article
Original Article
Exp Neurobiol 2019; 28(3): 376-389
Published online June 26, 2019
https://doi.org/10.5607/en.2019.28.3.376
© The Korean Society for Brain and Neural Sciences
Metabolomic Analysis Identifies Alterations of Amino Acid Metabolome Signatures in the Postmortem Brain of Alzheimer's Disease
Yoon Hwan Kim1,2, Hyun Soo Shim3, Kyoung Heon Kim2, Junghee Lee4,5, Bong Chul Chung1, Neil W. Kowall4,5, Hoon Ryu3,4,5,6*, and Jeongae Lee1,6*
1MolecularRecognition Research Center, Korea Institute of Science and Technology, Seoul 02792, Korea.
2Department of Biotechnology, Graduate School, Korea University, Seoul 02792, Korea.
3Center for Neuroscience, Brain Science Institute, Korea Institute of Science and Technology, Seoul 02792, Korea.
4Veteran's Affairs Boston Healthcare System, Boston, MA 02130, USA.
5Boston University Alzheimer's Disease Center and Department of Neurology, Boston University School of Medicine, Boston, MA 02118, USA.
6Convergence Research Center for Diagnosis, Treatment and Care System of Dementia, Korea Institute of Science and Technology, Seoul 02792, Korea.
Correspondence to: *To whom correspondence should be addressed.
Jeongae Lee, TEL: 82-2-958-6632, FAX: 82-2-958-5059
e-mail: frans@kist.re.kr
Hoon Ryu, TEL: 1-857-364-5910, FAX: 1-857-364-4540
e-mail: hoonryu@bu.edu
Abstract
Despite significant advances in neuroscience research over the past several decades, the exact cause of AD has not yet fully understood. The metabolic hypothesis as well as the amyloid and tau hypotheses have been proposed to be associated with AD pathogenesis. In order to identify metabolome signatures from the postmortem brains of sporadic AD patients and control subjects, we performed ultra performance liquid chromatography coupled with linear ion trap-Orbitrap mass spectrometer (UPLC-LTQ–Orbitrap-MS). Not only our study identified new metabolome signatures but also verified previously known metabolome profiles in the brain. Statistical modeling of the analytical data and validation of the structural assignments discovered metabolic biomarkers associated with the AD pathogenesis. Interestingly, hypotaurin, myo-inositol and oxo-proline levels were markedly elevated in AD while lutamate and N-acetyl-aspartate were decreased in the postmortem brain tissue of AD patients. In addition, neurosteroid level such as cortisol was significantly increased in AD. Together, our data indicate that impaired amino acid metabolism is associated with AD pathogenesis and the altered amino acid signatures can be useful diagnostic biomarkers of AD. Thus, modulation of amino acid metabolism may be a possible therapeutic approach to treat AD.
Graphical Abstract
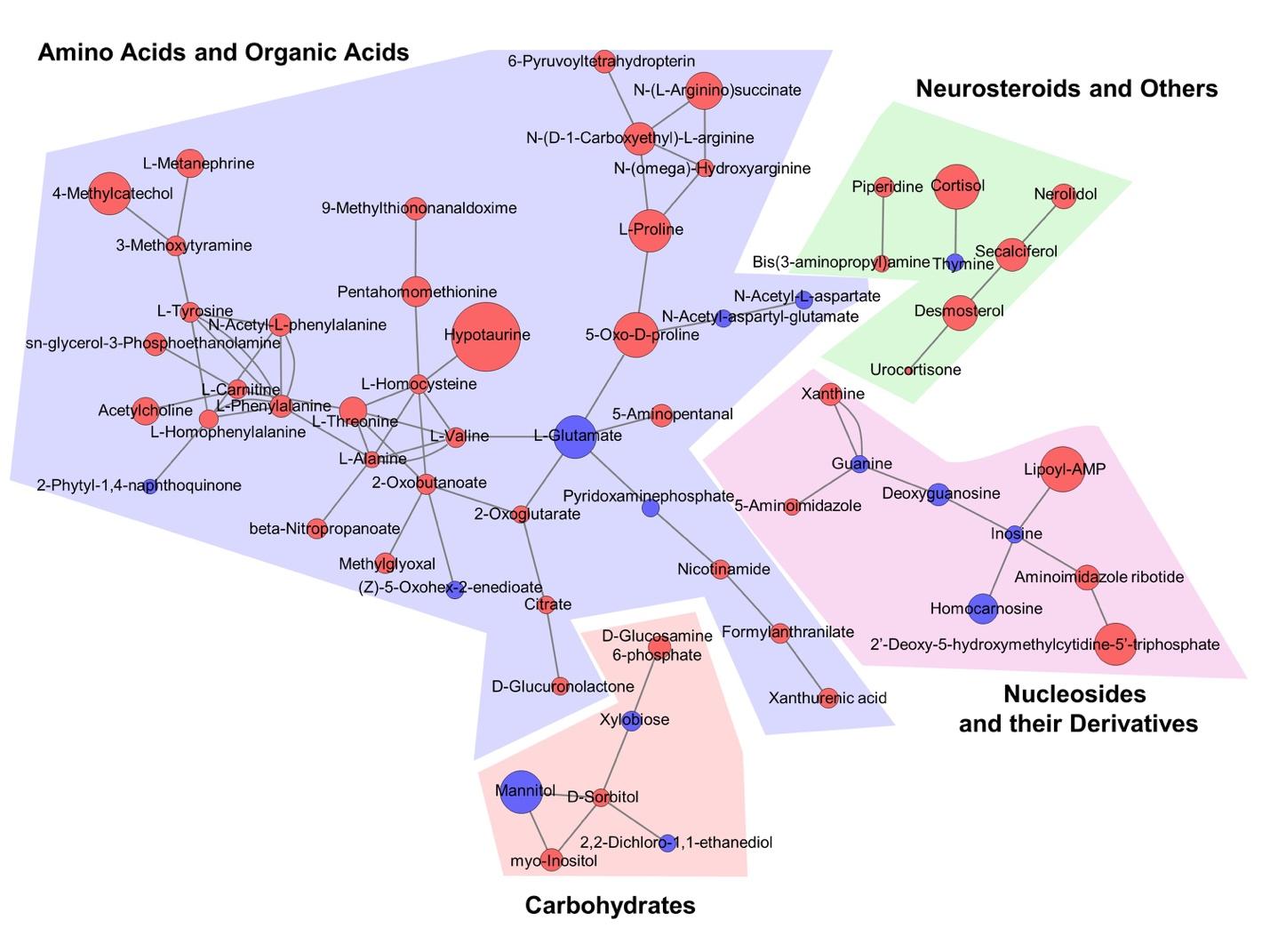
Keywords: Alzheimer's disease, Metabolomics, Liquid chromatography mass spectrometry, Biomarkers, amino acid metabolism


