Articles
Article Tools
Stats or Metrics
Article
Original Article
Exp Neurobiol 2019; 28(6): 679-696
Published online December 31, 2019
https://doi.org/10.5607/en.2019.28.6.679
© The Korean Society for Brain and Neural Sciences
Glial Cell Line-derived Neurotrophic Factor-overexpressing Human Neural Stem/Progenitor Cells Enhance Therapeutic Efficiency in Rat with Traumatic Spinal Cord Injury
Kyujin Hwang1,2†, Kwangsoo Jung3†, Il-Sun Kim2, Miri Kim2, Jungho Han3, Joohee Lim3, Jeong Eun Shin3, Jae-Hyung Jang4 and Kook In Park1,2,3*
1Brain Korea 21 Plus Project for Medical Science, Yonsei University College of Medicine, 2Yonsei Biomedical Research Institute, Yonsei University College of Medicine, 3Department of Pediatrics, Severance Children’s Hospital, Yonsei University College of Medicine, Seoul 03722, Korea, 4Department of Chemical and Biomolecular Engineering, Yonsei University, Seoul 03722, Korea
Correspondence to: *To whom correspondence should be addressed.
TEL: 82-10-9091-9231, FAX: 82-2-393-9118
e-mail: kipark@yuhs.ac
†These authors contributed equally.
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License
(http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and
reproduction in any medium, provided the original work is properly cited.
Abstract
Spinal cord injury (SCI) causes axonal damage and demyelination, neural cell death, and comprehensive tissue loss, resulting in devastating neurological dysfunction. Neural stem/progenitor cell (NSPCs) transplantation provides therapeutic benefits for neural repair in SCI, and glial cell line-derived neurotrophic factor (GDNF) has been uncovered to have capability of stimulating axonal regeneration and remyelination after SCI. In this study, to evaluate whether GDNF would augment therapeutic effects of NSPCs for SCI, GDNF-encoding or mock adenoviral vector-transduced human NSPCs (GDNF-or Mock-hNSPCs) were transplanted into the injured thoracic spinal cords of rats at 7 days after SCI. Grafted GDNF-hNSPCs showed robust engraftment, long-term survival, an extensive distribution, and increased differentiation into neurons and oligodendroglial cells. Compared with Mock-hNSPC- and vehicle-injected groups, transplantation of GDNF-hNSPCs significantly reduced lesion volume and glial scar formation, promoted neurite outgrowth, axonal regeneration and myelination, increased Schwann cell migration that contributed to the myelin repair, and improved locomotor recovery. In addition, tract tracing demonstrated that transplantation of GDNF-hNSPCs reduced significantly axonal dieback of the dorsal corticospinal tract (dCST), and increased the levels of dCST collaterals, propriospinal neurons (PSNs), and contacts between dCST collaterals and PSNs in the cervical enlargement over that of the controls. Finally grafted GDNF-hNSPCs substantially reversed the increased expression of voltage-gated sodium channels and neuropeptide Y, and elevated expression of GABA in the injured spinal cord, which are involved in the attenuation of neuropathic pain after SCI. These findings suggest that implantation of GDNF-hNSPCs enhances therapeutic efficiency of hNSPCs-based cell therapy for SCI.
Graphical Abstract
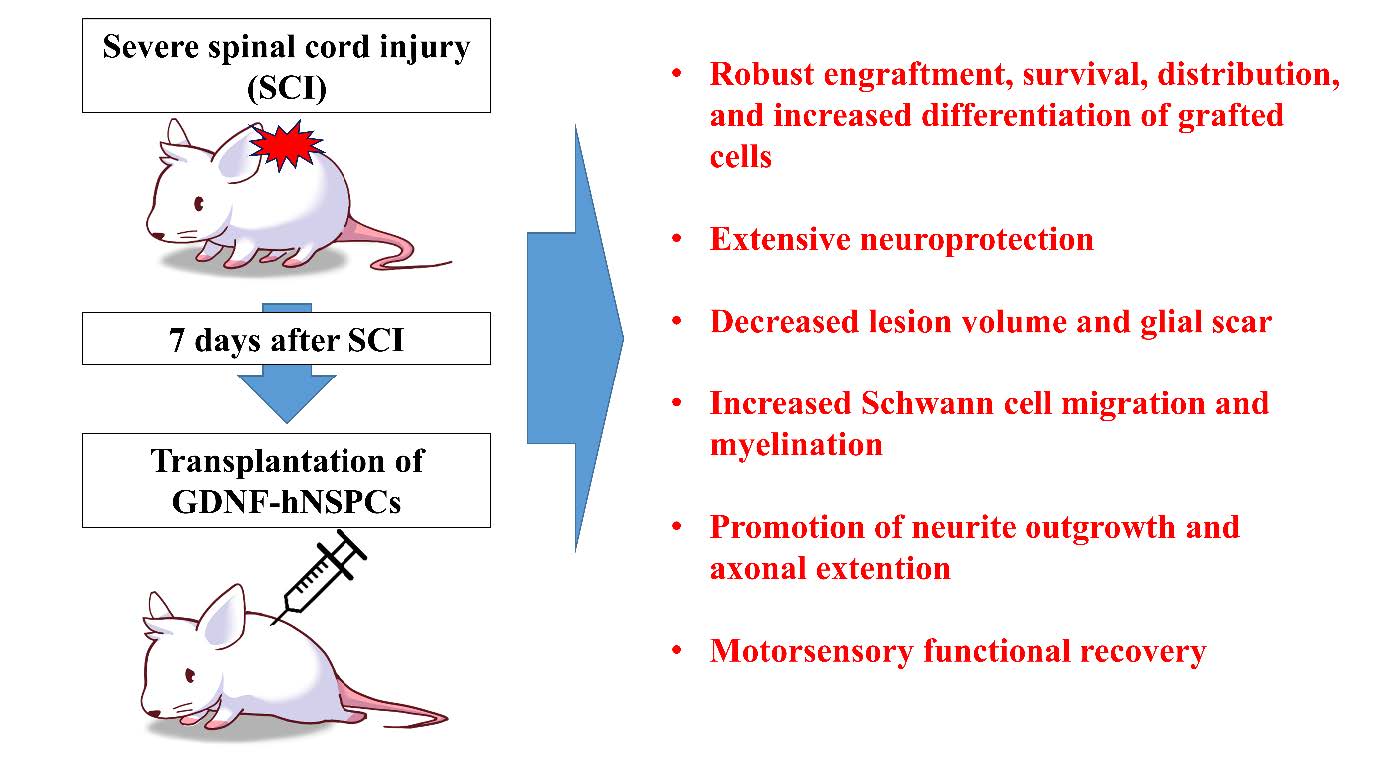
Keywords: Spinal cord injuries, Glial cell line-derived neurotrophic factor, Neural stem/progenitor cells, Cell-based therapy, Mechanical allodynia, Paraplegia


