Articles
Article Tools
Stats or Metrics
Article
Original Article
Exp Neurobiol 2021; 30(3): 222-231
Published online May 28, 2021
https://doi.org/10.5607/en21013
© The Korean Society for Brain and Neural Sciences
Adenovirus-induced Reactive Astrogliosis Exacerbates the Pathology of Parkinson’s Disease
Heeyoung An1,2,3, Hyowon Lee2,4, Seulkee Yang2,5, Woojin Won1,3, C. Justin Lee1,3* and Min-Ho Nam2,6*
1KU-KIST Graduate School of Converging Science and Technology, Korea University, Seoul 02841, 2Brain Science Institute, Korea Institute of Science and Technology (KIST), Seoul 02792, 3Center for Cognition and Sociality, Institute for Basic Science, Daejeon 34126, 4Department of Korean Medical Science, Graduate School, Kyung Hee University, Seoul 02447, 5Division of Bio-Medical Science & Technology, KIST School, University of Science and Technology, Seoul 02792, 6Department of KHU-KIST Convergent Science and Technology, Kyung Hee University, Seoul 02447, Korea
Correspondence to: *To whom correspondence should be addressed.
Min-Ho Nam, TEL: 82-2-958-6421, FAX: 82-2-958-7034
e-mail: dr.namminho@kist.re.kr
C. Justin Lee, TEL: 82-42-878-9150, FAX: 82-42-878-9151
e-mail: cjl@ibs.re.kr
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
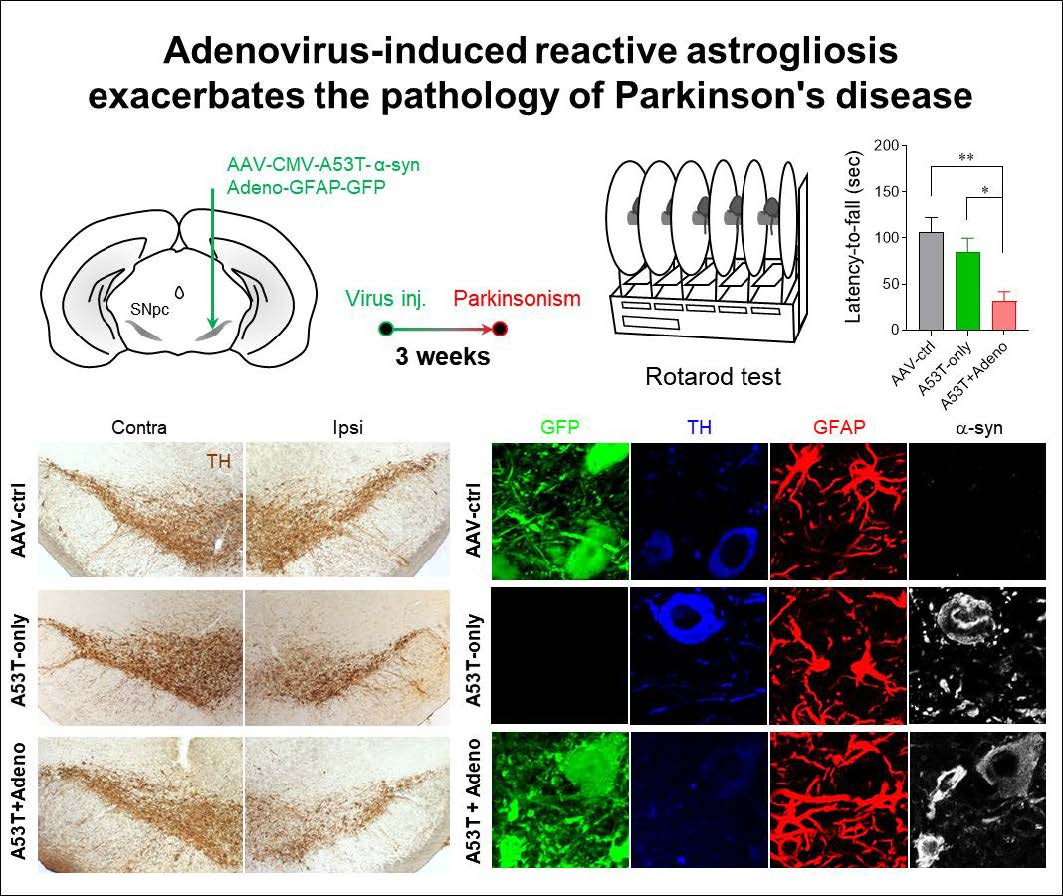
Parkinson’s disease (PD) is the most prevalent neurodegenerative motor disorder. While PD has been attributed to dopaminergic neuronal death in substantia nigra pars compacta (SNpc), accumulating lines of evidence have suggested that reactive astrogliosis is critically involved in PD pathology. These pathological changes are associated with α-synuclein aggregation, which is more prone to be induced by an A53T mutation. Therefore, the overexpression of A53T-mutated α-synuclein (A53T-α-syn) has been utilized as a popular animal model of PD. However, this animal model only shows marginal-to-moderate extents of reactive astrogliosis and astrocytic α-synuclein accumulation, while these phenomena are prominent in human PD brains. Here we show that Adeno-GFAP-GFP virus injection into SNpc causes severe reactive astrogliosis and exacerbates the A53T- α-syn-mediated PD pathology. In particular, we demonstrate that AAV-CMV-A53T-α-syn injection, when combined with Adeno-GFAP-GFP, causes more significant loss of dopaminergic neuronal tyrosine hydroxylase level and gain of astrocytic GFAP and GABA levels. Moreover, the combination of AAV-CMV-A53T-α-syn and Adeno-GFAP-GFP causes an extensive astrocytic α-syn expression, just as in human PD brains. These results are in marked contrast to previous reports that AAV-CMV-A53T-α-syn alone causes α-syn expression mostly in neurons but rarely in astrocytes. Furthermore, the combination causes a severe PD-like motor dysfunction as assessed by rotarod and cylinder tests within three weeks from the virus injection, whereas Adeno-GFAP-GFP alone or AAV-CMV-A53T-α-syn alone does not. Our findings implicate that inducing reactive astrogliosis exacerbates PD-like pathologies and propose the virus combination as an advanced strategy for developing a new animal model of PD.
Graphical Abstract
Keywords: Adenovirus infections, Parkinson’s disease, Alpha-synuclein, Reactive astrogliosis, Mouse model


