Articles
Article Tools
Stats or Metrics
Article
Original Article
Exp Neurobiol 2016; 25(1): 24-32
Published online February 29, 2016
https://doi.org/10.5607/en.2016.25.1.24
© The Korean Society for Brain and Neural Sciences
Agmatine Ameliorates High Glucose-Induced Neuronal Cell Senescence by Regulating the p21 and p53 Signaling
Juhyun Song1, Byeori Lee1, Somang Kang1,2, Yumi Oh1,2, Eosu Kim3, Chul-Hoon Kim4, Ho-Taek Song5 and Jong Eun Lee1,2*
1Department of Anatomy, Yonsei University College of Medicine, 2BK21 Plus Project for Medical Sciences, and Brain Research Institute, Yonsei University College of Medicine, 3Department of Psychiatry, Yonsei University College of Medicine, 4Department of Pharmacology, Yonsei University College of Medicine, 5Department of Diagnostic Radiology, Yonsei University College of Medicine, Seoul 03722, Korea
Correspondence to: *To whom correspondence should be addressed.
TEL: 82-2-2228-1646, 1659, FAX: 82-2-365-0700
e-mail: jelee@yuhs.ac
Neuronal senescence caused by diabetic neuropathy is considered a common complication of diabetes mellitus. Neuronal senescence leads to the secretion of pro-inflammatory cytokines, the production of reactive oxygen species, and the alteration of cellular homeostasis. Agmatine, which is biosynthesized by arginine decarboxylation, has been reported in previous
Keywords: Agmatine, High glucose, Hyperglycemia, Cell death, Senescence, Pro-inflammatory cytokines, p53, p21
INTRODUCTION
Diabetes is a growing public health concern with the increase in the worldwide population [1]. Hyperglycemia can lead to inflammation-induced metabolic disorders and various catabolism dysfunctions [2]. Hyperglycemia causes an acceleration of age-related damage of neurons [3]. Cellular senescence is defined as an irreversible proliferation arrest and contributes to age-associated decline in cellular homeostasis in diverse tissue [4]. Age-related pathologies are accompanied by an increase in proinflammatory cytokines, including tumor necrosis factor (TNF)-alpha and interleukin (IL)-6 [5]. Specifically, the release of IL-6, IL-1, TNF-alpha, and chemokine (C-C motif) ligand 2 (CCL2) has been reported to be involved in p53-mediated cellular senescence [6]. p53 is as a transcription factor that mediates cell cycle arrest, cell senescence, and apoptosis [7]. In the central nervous system (CNS), p53 induction has been shown to correlate with neuronal death and DNA damage and senescence [8] in
Agmatine is a cationic polyamine peptide that is synthesized by decarboxylation of L-arginine by arginine decarboxylase (ADC) [13]. Results of experimental studies have shown that agmatine has neuroprotective effects in CNS disorders including cerebral ischemia [14], Alzheimer's disease [15] and Parkinson's disease [16]. In diabetic rats with middle cerebral artery occlusion, posttreatment with agmatine reduces neurobehavioral dysfunction [17]. Moreover, DNA fragmentation and expression of proapoptotic proteins such as cleaved caspase-3 were significantly reduced in ADC-transfected neural stem cells against H2O2 stress [18]. Given that amine peptides are linked with the cell senescence through p53 and p21 cascade, we hypothesized that agmatine may alleviate the cell senescence by regulating p21 and p53 signaling. Here, we aimed to study the effects of agmatine on high glucose-induced neuronal cellular damage, with a focus on the p21 and p53 pathway, to test the hypothesis that agmatine could reverse high glucose-induced cellular senescence.
MATERIALS AND METHODS
N2A cells possess some of the properties of neuronal stem cells and are capable of differentiating into neuron-like cells in the presence of retinoic acid (RA) (Sigma-Aldrich, St. Louis, MO, USA). Undifferentiated N2A cells were cultured in Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% fetal bovine serum (FBS; Gibco, Grand Island, NY, USA) and 100 µg/mL penicillin-streptomycin (Gibco, Grand Island, NY, USA). N2A cells were passaged at least twice and then plated at 1×104 cells/mL in DMEM supplemented with 10% FBS for 24 hours. After that, the medium was changed to DMEM supplemented with 1% FBS and 5 µM RA for differentiation. Cultures were maintained in a humidified atmosphere of 5% CO2 at 37℃. Cells were treated with D-glucose (Sigma-Aldrich, St. Louis, MO, USA) and agmatine (100 µM) (Sigma-Aldrich, St. Louis, MO, USA) for 24 hours before sampling. As several in vitro studies [19], we experimented in the concentration of glucose (25~200 mM).
N2A cells were seeded at 1×104 cells/mL in 96-well plates to examine the effects of all experimental treatments. Cells were then rinsed with phosphate-buffered saline (PBS), and culture medium was replaced with serum-free medium. Next, 100 µl 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) (Sigma, St. Louis, MO, USA) solution (5 mg/ml in PBS) was added per well. After 1 hour 30 min of incubation, the medium was removed, and dimethyl sulfoxide was added to solubilize the purple formazan product of the MTT reaction. The supernatant from each well was measured at a wavelength of 570 nm with background subtraction at 650 nm. All experiments were repeated at least 5 times. Cell viability was expressed as a percentage relative to the normal control group value.
The level of ROS in N2A cells was measured using a fluorescent probe, 2' 7'-dichlorodihydrofluorescein diacetate (DCF-DA; Invitrogen, Carlsbad, CA, USA), as previously described [20]. N2A cells were passaged at least twice and then plated at 1 × 104 cells/mL in DMEM supplemented with 10% FBS for 24 hours. After that, the medium was changed to DMEM supplemented with 1% FBS and 5 µM RA for differentiation. Cells were treated with glucose and agmatine for 24 hours. Then, N2A cells were treated with 5 µM DCF-DA for 30 min at 37℃. After washing with PBS, fluorescence was measured with a microscope (Olympus, Korea) equipped with a CCD camera (Hamamatsu Photonics, Japan).
After agmatine treatment and exposure to high glucose stress, N2A cells were washed twice with ice-cold PBS, scraped, and collected. N2A cell pellets were lysed with ice-cold RIPA buffer (Sigma-Aldrich, St. Louis, MO, USA). The lysates were centrifuged at 16,100g for 1 hour at 4℃ to produce whole-cell extracts. Protein content was quantified using the BCA protein assay kit (Pierce, IL, USA). Protein (35 µg) was separated on a 10% SDS-polyacrylamide gel and transferred onto a polyvinylidene difluoride membrane. After blocking with 5% bovine serum albumin in TBS/Tween (20 nM Tris (pH 7.2), 150 mM NaCl, 0.1% Tween 20) for 1 hour at room temperature, immunoblots were incubated overnight at 4℃ with primary antibodies that specifically detect cleaved caspase 3 (1:2000, Santa Cruz Biotechnology, Santa Cruz, CA, USA), caspase 3 (1:2000, Abcam, Cambridge, MA, USA), phosphor-p53 (1:2000, Abcam, Cambridge, MA, USA), p53 (1:2000, Abcam, Cambridge, MA, USA), or β-actin (1:2000, Cell Signaling, Danvers, MA, USA). Blots were then incubated with horseradish peroxidase-linked anti-mouse or -rabbit IgG antibodies (Abcam, Cambridge, MA, USA) for 2 hours at room temperature. Protein bands were detected and analyzed using enhanced chemiluminescence (ECL; Pierce, IL, USA).
The expression of Bax, TNF-alpha, CCL2, p21, cyclin A, PARP-1, p35, Cdk 5 in N2A cells treated with high glucose and agmatine was examined using reverse transcription PCR with the corresponding primers. Samples were lysed with Trizol reagent (Invitrogen, Carlsbad, CA, USA). PCR was performed using the following thermal cycling conditions: 10 min at 95℃; 35 cycles of denaturing at 95℃ for 15 sec; annealing at 63℃for 30 sec and elongation at 72℃ for 30 sec; final extension at 72℃ for 10 min; and holding at 4℃. PCR was performed using the following primers (5' to 3'): TNF-alpha (F) CAA GGG ACA AGG CTG CCC CG, (R) GCA GGG GCT CTT GAC GGC AG, CCL2 (F) CTC GAG ATG CAG GTC CCT GTC AT, (R) AAG CTT CTA GTT CAC TGT CAC ACT G, p21 (F) AGT GTG CCG TTG TCT CTT CG, (R) ACA CCA GAG TGC AAG ACA GC, Cyclin A (F) AGT ACC TGC CTT CAC TCA TTG CTG, (R) TCT GGT GAA GGT CCA CAA GAC AAG, PARP-1 (F) AGG CCC TAA AGG CTC AGA AT, (R) CTA GGT TTC TGT GTC TTG AC, Cdk 5 (F) GGC TAA AAA CCG GGA AAC TC, (R) CCA TTG CAG CTG TCG AAA TA, p35 (F) AAG AAC GCC AAG GAC AAG AA, (R) TCA TTG TTG AGG TGC GTG AT, β-actin (F) TCT GGC ACC ACA CCT TCT A, (R) AGG CAT ACA GGG ACA GCA C. PCR products were separated by gel electrophoresis using 1.5% agarose gels and stained with ethidium bromide.
To examine the amount of IL-6 mRNA in conditioned N2A cells, quantitative real time-PCR was performed using IL-6 primers. A One Step SYBR® Prime Script TM RT-PCR Kit II (Takara, Japan) was used to conduct qRT-PCR. PCR was performed using the following primers (5' to 3'): IL-6 (F) AAC GAT GA TGC ACT TGC AGA, (R) CTC TGA AGG ACT CTG GCT TTG, β-actin (F) TCT GGC ACC ACA CCT TCT A, (R) AGG CAT ACA GGG ACA GCA C. Denaturing was carried out at 95℃ for 3 min; 40 cycles of 95℃ for 20 sec; annealing at 60℃ for 20 sec; and extension at 72℃ for 20 sec. At each extension step at 72℃, fluorescence was detected at 585 nm. The expression of IL-6 was assessed using an ABI prism 7500 Real-Time PCR System (Life Technologies Corporation, CA, USA) and analyzed with comparative Ct quantification. β-actin was amplified as an internal control. The ΔCt values of high glucose-exposed N2A cells were compared with the ΔCt values of normal N2A cells.
N2A cells under the experimental condition were fixed in 0.2% glutaraldehyde solution for 15 min at room temperature. To detect β-galactosidase activation, we were performed β-galactosidase staining (Cell Signaling Technology, Beverly, MA, USA) at pH 6.0 [21]. For quantification, approximately five randomly chosen microscope fields in three independent cultures were examined at ×20 magnification so that at least 30 cells were considered as being either β-galactosidase positive or negative. β-galactosidase positive had cytoplasmic staining that appeared dark blue. All the experiments were repeated 3 times. N2A cells were then visualized under a confocal microscope (Zeiss LSM 700, Carl Zeiss, Oberkochen, Germany).
Statistical analyses were conducted using SPSS 18.0 software (IBM Corp., Armonk, NY, USA). Data are expressed as the mean±standard error of the mean of three independent experiments. The statistical significance of group differences was determined by one-way analysis of variance (ANOVA) followed by Bonferroni post hoc multiple comparison tests. Differences compared with normal control were considered statistically significant at *p<0.05, **p<0.001 and differences within groups were marked using lines and drew * and ** to show significancy.
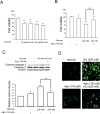
To examine the effect of high glucose stress on neuronal cell, we conducted a cell viability assay (Fig. 1A and 1B), western blot analysis (Fig. 1C), DCF-DA assay (Fig. 1D). In the cell viability assay, we observed markedly decreased cell viability depending on glucose concentration compared to control treatment (Fig. 1A). Based on MTT assay, we choose 200 mM of glucose as a high glucose stress condition because it induces high glucose stress as much as possible but not causes severe cell death. Fig. 1B showed the agmatine's effect on the cell viability under glucose 200 mM treatment (Fig. 1B). The increase in cleaved caspase-3 protein levels induced by high glucose condition was reduced by agmatine (Fig. 1C). ROS generation was measured using DCF-DA reagent in conditioned N2A cells (Fig. 1D). High glucose (200 mM) treatment induced significantly ROS generation in N2A cells (Fig. 1D). In comparison, agmatine attenuated the generation of ROS in N2A (Fig. 1D) cells in high glucose stress condition. Given our results, we hypothesize that agmatine may rescue neuronal cells from cell death under high glucose conditions.
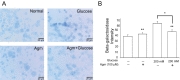
To determine the β-galactosidase as a hydrolase enzyme which accumulated in senescence cells under high glucose, we conducted β-Galactosidase staining (Fig. 2A and 2B). By comparison with the normal control group, β-galactosidase positive cells in high glucose condition were observed more positive cells (Fig. 2A and 2B). In contrast to the increased β-Galactosidase positive cells (blue) in high glucose group, β-Galactosidase positive cells were consciously reduced by agmatine treatment (Fig. 2B). Judging from β-galactosidase was expressed in senescence cells, our findings indicate that high glucose generates the cellular senescence and agmatine protects the cellular senescence against high glucose stress.
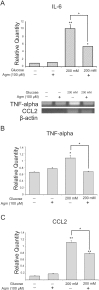
To estimate the alteration of inflammatory cytokine expression in neuronal cells exposed to high glucose conditions, we conducted real-time PCR (Fig. 3A) and reverse transcription PCR (Fig. 3B and 3C). Under high glucose condition, the mRNA expression of pro-inflammatory cytokines such as IL-6 (Fig. 3A), TNF-alpha (Fig. 3B), and CCL2 (Fig. 3C) was increased. However, agmatine treatment reduced the high glucose-induced increase in IL-6 (Fig. 3A), TNF-alpha (Fig. 3B), and CCL2 (Fig. 3C) mRNA levels compared to neuronal cells exposed to high glucose condition alone. Our results showed that increased expression of pro-inflammatory cytokines caused by high glucose was reduced by agmatine.
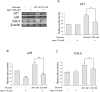
To investigate p21 signaling related to cellular senescence in neuronal cells under the high glucose condition, we performed reverse transcription PCR (Fig. 4). In the high glucose condition, mRNA levels of p21 (Fig. 4A) increased with regard to cellular senescence in comparison with the normal control group (Fig. 4A). Agmatine treatment reduced the increase in mRNA levels of p21 in the high glucose-exposed N2A cells (Fig. 4A). Moreover, the mRNA levels of p35 (Fig. 3B), Cdk 5 (Fig. 4C), were significantly increased in the high glucose condition, and were attenuated by agmatine treatment (Fig. 4B and 4C). In this regard, agmatine may inhibit neuronal cell senescence via p21-mediated cell senescence signaling in hyperglycemia.
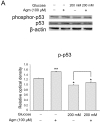
To assess the phosphorylation of p53 in N2A cells, we checked the protein level of phosphorylated p53 using western blotting. Agmatine results in the increase of p53 phosphorylation (Fig. 5C) with regard to cellular senescence in comparison with the normal control group (Fig. 5C). In high glucose condition, agmatine increased phosphorylation of p53 in the N2A cells (Fig. 5C). Taken together, agmatine may inhibit neuronal cell senescence by activating p53 in hyperglycemia.
Hyperglycemia leads to cell death, oxidative stress and cellular senescence, which is defined as an irreversible cessation of mitosis via p21-dependent signaling that occurs in the early stages of diabetic nephropathy [11]. In the cell, caspase 3 activation showed the cell death in response to the stress. In the present study, decrease of cleaved caspase 3 expression in high glucose conditioned N2A cells by agmatine indicates that agmatine may rescue the cells against the high glucose-induced cell death (Fig. 1C). Several researches demonstrated that the cellular senescence is commonly characterized by high β-galactosidase activity [22]. In many researches, β-galactosidase activity has been used as the important marker of cell senescence
Neuronal senescence results in cytokine dysregulation with an increase of pro-inflammatory cytokines and reduction of anti-inflammatory cytokines, leading to neurodegenerative diseases. Increased levels of TNF-alpha, IL-1, and IL-6 aggravate the process of neurodegenerative pathology [5]. Some studies demonstrated that cell senescence via the p21 pathway causes an increase in pro-inflammatory cytokine levels in patients [5]. Specifically, pro-inflammatory cytokines TNF-α [25], IL-6 [26], and CCL2 [27] have been shown to increase excitotoxic neuronal damage. In present study, our results assume that agmatine may attenuate the high glucose-induced increase of pro-inflammatory cytokines such as IL-6 (Fig. 3A), TNF-alpha (Fig. 3B), and CCL2 (Fig. 3C) through inhibition of p21-mediated signaling, suggesting that these pro-inflammatory cytokines were regulated by the p21 pathway.
The activation of p53 has been reported that it could promote cell cycle arrest and DNA repair [7]. In addition, p53 activates numerous anti-oxidant genes such as
Consequently, in present study, our results indicate that agmatine alleviates the neuronal cell senescence by high glucose by downregulaing p21 and by activating p53.
Thus, in accordance with our findings, we propose that further research is needed to elucidate that the effect of agmatine on the p21 and p53-medaiated signaling, which may serve as a target for the development of promising therapeutic strategies in diabetes-induced neuropathy.
- Cowie CC, Rust KF, Ford ES, Eberhardt MS, Byrd-Holt DD, Li C, Williams DE, Gregg EW, Bainbridge KE, Saydah SH, Geiss LS. Full accounting of diabetes and pre-diabetes in the U.S. population in 1988-1994 and 2005-2006. Diabetes Care 2009;32:287-294.
- Senn JJ, Klover PJ, Nowak IA, Mooney RA. Interleukin-6 induces cellular insulin resistance in hepatocytes. Diabetes 2002;51:3391-3399.
- Smith-West C, Garris DR. Diabetes-associated hypothalamic neuronal depopulation in the aging Chinese hamster. Brain Res 1983;285:385-389.
- Campisi J. Senescent cells, tumor suppression, and organismal aging: good citizens, bad neighbors. Cell 2005;120:513-522.
- Licastro F, Pedrini S, Caputo L, Annoni G, Davis LJ, Ferri C, Casadei V, Grimaldi LM. Increased plasma levels of interleukin-1, interleukin-6 and alpha-1-antichymotrypsin in patients with Alzheimer's disease: peripheral inflammation or signals from the brain?. J Neuroimmunol 2000;103:97-102.
- Noureddine H, Gary-Bobo G, Alifano M, Marcos E, Saker M, Vienney N, Amsellem V, Maitre B, Chaouat A, Chouaid C, Dubois-Rande JL, Damotte D, Adnot S. Pulmonary artery smooth muscle cell senescence is a pathogenic mechanism for pulmonary hypertension in chronic lung disease. Circ Res 2011;109:543-553.
- Kruse JP, Gu W. Modes of p53 regulation. Cell 2009;137:609-622.
- Sui X, Han W, Pan H. p53-induced autophagy and senescence. Oncotarget 2015;6:11723-11724.
- Goldstein I, Yizhak K, Madar S, Goldfinger N, Ruppin E, Rotter V. p53 promotes the expression of gluconeogenesisrelated genes and enhances hepatic glucose production. Cancer Metab 2013;1:9.
- Masgras I, Carrera S, de Verdier PJ, Brennan P, Majid A, Makhtar W, Tulchinsky E, Jones GD, Roninson IB, Macip S. Reactive oxygen species and mitochondrial sensitivity to oxidative stress determine induction of cancer cell death by p21. J Biol Chem 2012;287:9845-9854.
- Kitada K, Nakano D, Ohsaki H, Hitomi H, Minamino T, Yatabe J, Felder RA, Mori H, Masaki T, Kobori H, Nishiyama A. Hyperglycemia causes cellular senescence via a SGLT2- and p21-dependent pathway in proximal tubules in the early stage of diabetic nephropathy. J Diabetes Complications 2014;28:604-611.
- Mao D, Hinds PW. p35 is required for CDK5 activation in cellular senescence. J Biol Chem 2010;285:14671-14680.
- Tabor CW, Tabor H. Polyamines. Annu Rev Biochem 1984;53:749-790.
- Gilad GM, Salame K, Rabey JM, Gilad VH. Agmatine treatment is neuroprotective in rodent brain injury models. Life Sci 1996;58:PL 41-PL 46.
- Song J, Hur BE, Bokara KK, Yang W, Cho HJ, Park KA, Lee WT, Lee KM, Lee JE. Agmatine improves cognitive dysfunction and prevents cell death in a streptozotocininduced Alzheimer rat model. Yonsei Med J 2014;55:689-699.
- Gilad GM, Gilad VH, Finberg JP, Rabey JM. Neurochemical evidence for agmatine modulation of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) neurotoxicity. Neurochem Res 2005;30:713-719.
- Cui H, Lee JH, Kim JY, Koo BN, Lee JE. The neuroprotective effect of agmatine after focal cerebral ischemia in diabetic rats. J Neurosurg Anesthesiol 2012;24:39-50.
- Bokara KK, Kwon KH, Nho Y, Lee WT, Park KA, Lee JE. Retroviral expression of arginine decarboxylase attenuates oxidative burden in mouse cortical neural stem cells. Stem Cells Dev 2011;20:527-537.
- Zhang J, Li B, Zheng Z, Kang T, Zeng M, Liu Y, Xia B. Protective effects of Notch1 signaling activation against high glucose-induced myocardial cell injury: analysis of its mechanisms of action. Int J Mol Med 2015;36:897-903.
- Royall JA, Ischiropoulos H. Evaluation of 2',7'-dichlorofluorescin and dihydrorhodamine 123 as fluorescent probes for intracellular H2O2 in cultured endothelial cells. Arch Biochem Biophys 1993;302:348-355.
- Burn SF. Detection of β-galactosidase activity: X-gal staining. Methods Mol Biol 2012;886:241-250.
- Serrano M, Lin AW, McCurrach ME, Beach D, Lowe SW. Oncogenic ras provokes premature cell senescence associated with accumulation of p53 and p16INK4a. Cell 1997;88:593-602.
- Waldman T, Kinzler KW, Vogelstein B. p21 is necessary for the p53-mediated G1 arrest in human cancer cells. Cancer Res 1995;55:5187-5190.
- Chang BD, Watanabe K, Broude EV, Fang J, Poole JC, Kalinichenko TV, Roninson IB. Effects of p21Waf1/Cip1/Sdi1 on cellular gene expression: implications for carcinogenesis, senescence, and age-related diseases. Proc Natl Acad Sci U S A 2000;97:4291-4296.
- Goretsky T, Dirisina R, Sinh P, Mittal N, Managlia E, Williams DB, Posca D, Ryu H, Katzman RB, Barrett TA. p53 mediates TNF-induced epithelial cell apoptosis in IBD. Am J Pathol 2012;181:1306-1315.
- Volonte D, Zou H, Bartholomew JN, Liu Z, Morel PA, Galbiati F. Oxidative stress-induced inhibition of Sirt1 by caveolin-1 promotes p53-dependent premature senescence and stimulates the secretion of interleukin 6 (IL-6). J Biol Chem 2015;290:4202-4214.
- Tang X, Asano M, O'Reilly A, Farquhar A, Yang Y, Amar S. p53 is an important regulator of CCL2 gene expression. Curr Mol Med 2012;12:929-943.
- Kastan MB, Onyekwere O, Sidransky D, Vogelstein B, Craig RW. Participation of p53 protein in the cellular response to DNA damage. Cancer Res 1991;51:6304-6311.
- Buzzai M, Jones RG, Amaravadi RK, Lum JJ, DeBerardinis RJ, Zhao F, Viollet B, Thompson CB. Systemic treatment with the antidiabetic drug metformin selectively impairs p53-deficient tumor cell growth. Cancer Res 2007;67:6745-6752.
- Chae YB, Kim MM. Activation of p53 by spermine mediates induction of autophagy in HT1080 cells. Int J Biol Macromol 2014;63:56-63.



