Articles
Article Tools
Stats or Metrics
Article
Original Article
Exp Neurobiol 2023; 32(3): 147-156
Published online June 30, 2023
https://doi.org/10.5607/en23007
© The Korean Society for Brain and Neural Sciences
Monitoring α-synuclein Aggregation Induced by Preformed α-synuclein Fibrils in an In Vitro Model System
Beom Jin Kim1,2,3, Hye Rin Noh1,2,3, Hyongjun Jeon2,3 and Sang Myun Park1,2,3*
1Department of Pharmacology, Ajou University School of Medicine, Suwon 16499, 2Center for Convergence Research of Neurological Disorders, Ajou University School of Medicine, Suwon 16499, 3Neuroscience Graduate Program, Department of Biomedical Sciences, Ajou University School of Medicine, Suwon 16499, Korea
Correspondence to: *To whom correspondence should be addressed.
TEL: 82-31-219-5063, FAX: 82-31-219-5069
e-mail: sangmyun@ajou.ac.kr
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
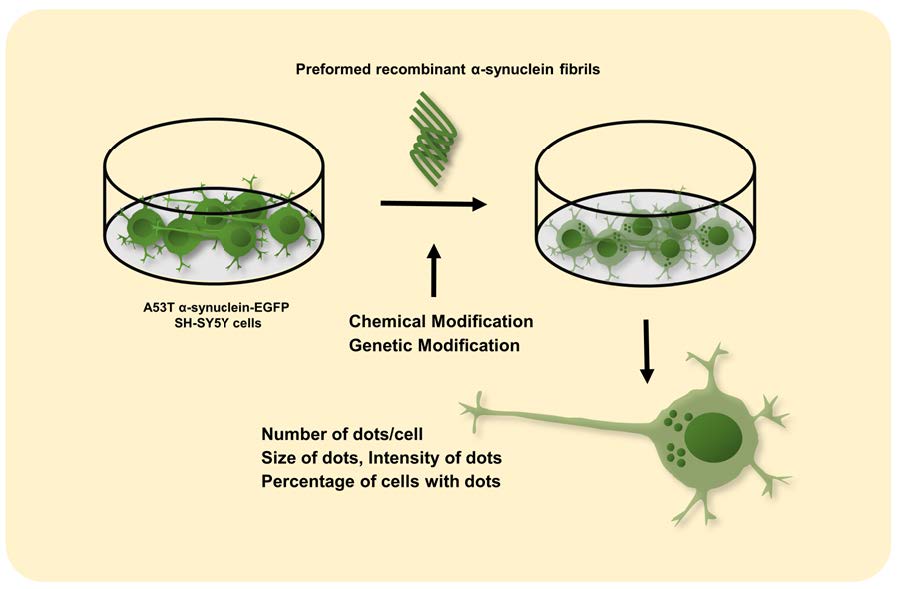
Parkinson’s disease (PD) is characterized by the presence of α-synuclein (α-syn) inclusions in the brain and the degeneration of dopamine-producing neurons. There is evidence to suggest that the progression of PD may be due to the prion-like spread of α-syn aggregates, so understanding and limiting α-syn propagation is a key area of research for developing PD treatments. Several cellular and animal model systems have been established to monitor α-syn aggregation and propagation. In this study, we developed an
Graphical Abstract
Keywords: Parkinson disease, α-synuclein, Protein aggregation,


