Articles
Article Tools
Stats or Metrics
Article
Technologue
Exp Neurobiol 2018; 27(1): 65-75
Published online February 28, 2018
https://doi.org/10.5607/en.2018.27.1.65
© The Korean Society for Brain and Neural Sciences
Development of a Low-cost, Comprehensive Recording System for Circadian Rhythm Behavior
Jea Kwon1,2,3, Min Gu Park1,2,3, Seung Eun Lee4 and C. Justin Lee1,2,3,*
1KU-KIST Graduate School of Converging Science and Technology, Korea University, Seoul 02841, 2Center for Neuroscienceand Functional Connectomics, Korea Institute of Science and Technology (KIST), Seoul 02792, 3Center for Glia-NeuronInteraction, Korea Institute of Science and Technology (KIST), Seoul 02792, 4Virus Facility, Research Animal Resource Center,Korea Institute of Science and Technology (KIST), Seoul 02792, Korea
Correspondence to: *To whom correspondence should be addressed.
TEL: 82-2-958-6940, FAX: 82-2-958-6937
e-mail: cjl@kist.re.kr
Abstract
Circadian rhythm is defined as a 24-hour biological oscillation, which persists even without any external cues but also can be re-entrained by various environmental cues. One of the widely accepted circadian rhythm behavioral experiment is measuring the wheel-running activity (WRA) of rodents. However, the price for commercially available WRA recording system is not easily affordable for researchers due to high-cost implementation of sensors for wheel rotation. Here, we developed a cost-effective and comprehensive system for circadian rhythm recording by measuring the house-keeping activities (HKA). We have monitored animal's HKA as electrical signal by simply connecting animal housing cage with a standard analog/digital converter: input to the metal lid and ground to the metal grid floor. We show that acquired electrical signals are combined activities of eating, drinking and natural locomotor behaviors which are well-known indicators of circadian rhythm. Post-processing of measured electrical signals enabled us to draw actogram, which verifies HKA to be reliable circadian rhythm indicator. To provide easy access of HKA recording system for researchers, we have developed user-friendly MATLAB-based software, Circa Analysis. This software provides functions for easy extraction of scalable “touch activity” from raw data files by automating seven steps of post-processing and drawing actograms with highly intuitive user-interface and various options. With our cost-effective HKA circadian rhythm recording system, we have estimated the cost of our system to be less than $150 per channel. We anticipate our system will benefit many researchers who would like to study circadian rhythm.
Graphical Abstract
Keywords: Circadian rhythm, Mice, Electrical Equipment and Supplies, Software
INTRODUCTION
Circadian rhythm is defined as a 24 hour biological rhythm, which persists even without any external cues but also can be re-entrained by various environmental cues [1,2,3]. Accumulating lines of evidence indicate that this biological rhythm is not only important for periodic daily behaviors of organisms, but also critically linked to various forms of pathophysiological processes including metabolism, immunity, neurodegeneration and cognitive function [4,5,6,7]. As a result, there has been a growing need of interdisciplinary research on circadian rhythm and other research areas. However, researchers who are newly getting into the fields of circadian rhythm soon face high cost setup fees for the behavioral experiments. For this reason, reducing financial burden for research equipment can be a good way to accelerate interdisciplinary research on circadian rhythm.
In the field of chronobiology, most circadian rhythm behavioral experiments have been based on animal's locomotor activity [8,9]. For rodents, the most widely accepted method for measuring circadian locomotor activity is wheel-running activity (WRA) recording system. However, cost for commercially available WRA configurations is not easily affordable for many researchers since these instruments come at a cost of tens of thousands of U.S. dollars to hundreds of thousands of U.S. dollars. The factors for high price in commercial WRA configurations could be divided into three categories: 1) custom-developed animal housing environment and cages with running wheel, 2) activity counting/recording devices and monitoring system, 3) high cost commercial analysis software. Since these systems are usually provided as a whole package, there are not many options for consumers to reduce the price.
In addition to high cost, there are also some intrinsic disadvantages of WRA configuration of currently available circadian rhythm recording systems. One of the critical issues on WRA configuration is that the usage of wheel-running behavior itself can alter the animal's endogenous activity patterns, energy balance, food intake, motivation and reward systems, stress, anxiety, depression and even brain structures [10,11,12,13,14,15]. Moreover, when an animal fails to learn wheel-running behaviors or is unable to utilize wheels because of innate motor defects, WRA configuration cannot measure the animal's circadian rhythm. Therefore, measuring the endogenous circadian rhythms with natural locomotor activities, such as drinking/feeding behaviors and sleep/wake cycles would be much more suitable for monitoring animal's inherent circadian rhythm.
There have been several approaches proposed to monitor natural activities, including vibration detecting system [16,17,18], home cage system [16,19,20], beam break system [16], video monitoring system [16,21], RFID recording system [16,22], and piezo-sensor system [23]. While these systems were successful in terms of detecting natural animal behaviors, none of these systems seems to achieve noticeable cost reduction compared to WRA system. In terms of cost, rather simple wheel & wheel-counting sensors are generally cheaper than any of sensors used in the above system. Therefore, currently available systems including WRA system are not capable of recording natural activities at a reasonable cost.
To overcome the issues with currently available systems, we developed a novel circadian rhythm recording system as an alternative option. We provide a novel cost-effective solution for circadian rhythm recording system which monitors natural activities. By simply connecting a metal lid of animal housing cage to an input channel of a standard analog/digital (A/D) converter by copper wire, we were able to record animal's movement when a mouse touches a metal lid while standing on a metal floor (ground), generating an electrical signal. This idea has been previously described as a low-cost solution to detect drinking behavior in mice with various additional advantages [24]. Since animal's food pellets and water bottle are generally placed above the metal lid of a cage, the recorded electrical signals from animal's “touching behavior” can reflect combined house-keeping activities (HKA) of eating, drinking and some of natural locomotor behavior such as grabbing the metal lid, which are all well-known indicators for circadian rhythm. To convert these electrical signals into scalable activity, we have developed MATLAB-based user-friendly software, Circa Analysis. This software is optimized for HKA recording system and can automatically draw actograms. In combination with a novel hardware configuration and optimized software, our system can achieve simultaneous recordings from 16 mice for duration of indefinite time. By eliminating the need for a running-wheel and commercial software, we were able to reduce the cost down to 1/10th of commercial systems.
MATERIALS AND METHODS
Adult male and female mice (>56 days old) from BALB/cByJ were housed in standard distributional type of housing cages in a temperature and humidity-controlled sound proof box on a 12:12h light-dark cycle for 1 week before the beginning of the experiment. The intensity of the LED light that mice experience at the bottom of the housing cage is adjusted to 300 lux. Humidity and temperature for animal maintaining chambers were kept between 40~60% and 25~26℃. All experimental procedures were conducted according to protocols approved by the directives of the Institutional Animal Care and Use Committee of the Institutional Animal Care and Use Committee (Approval number :201603025) of KIST (Seoul, Republic of Korea).
For animal housing floor material, metal grid floor was optimal for long term recording compared to flat metal floor, although both materials provided similar conduction, resistance, and accuracy of electrical signals. This is because mice feces can be easily accumulated in flat metal floor. Moreover, flat metal floor has been shown to hamper adequate footing of animals. Therefore, metal grid floor is suitable for long term recording, considering animal's welfare.
For animal housing cages, we used metal grid floor type distributional mouse cage with metal lid (JEUNG DO Bio & Plant, Korea) (Fig. 1A). Sound proof LED light-dark chamber was custom ordered from the company (JEUNG DO Bio & Plant, Korea). For standard A/D converter, we have utilized a Digidata 1440a (Molecular Device) for simultaneous 16 channel recording. The recording environment of computer was Intel G2020 2.90 GHz dual core processer with 2 GB RAM and 1.5 TB hard drive running on Windows 7 OS. Our Circa Analysis software was developed on MATLAB version 2017b. For recording software, Axoscope ver. 10.6 (freely available from Molecular Device) was used. With our HKA system, it was possible to record up to 16 channel simultaneously with A/D converter of Digidata 1440a model (Up to 8 channels with 1320a model). Circa analysis software provides up to 16 channel simultaneous analysis.


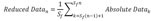
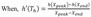
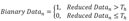
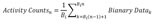
*HPF: High Pass Filter, Butterworth 5th order, 1 Hz cut-off
*Rosin's: Rosin's thresholding algorithm
*
*
*
*
The A/D converter used in our HKA system has 16-bit resolution, 16 input channels, 1 to 250 kHz sampling rate range, −10.00 to +10.00 V of input dynamic range, 1MOhm of input resistance. Since the sampling frequency of our recording condition was 20Hz. We recorded 16 channel simultaneously, therefore the precise calculation of the single data size is 52.7 MB.
16(bit)×20 (Hz)×86,400 (second)×16 (channel)=442,368,000 (bit)
442,368,000 (bit)×0.125=55,296,000 (byte)
55,296,000 (byte)÷106=52.7 (MB)
RESULTS
A previous study has shown that simply connecting standard A/D converter with metal sipper tube of water bottle and metal grid floor can precisely detect animal's licking behavior [24]. In detail, a junction potential is generated whenever animal licks the tube and closes the electrical circuit for the duration of the tongue-sipper tube contact [24]. Junction potentials occur wherever dissimilar conductors are in contact. Based on this idea, we hypothesized that connecting the metal grid floor and the metal lid of the animal housing cage would generate electrical signals composed of any lid-touching behaviors. To test this hypothesis, we have plugged one side of BNC cable with input of the standard A/D converter and another side with animal's housing cage, by connecting BNC core part at the metal lid and BNC shell part at the metal grid floor (Fig. 1A). Next, we placed laboratory mouse inside the cage and monitored animal's behavior and signal generation. The signals are generated when animals grab, eat and drink (Fig. 1C). Whenever animals are just crawling the floor, hanging on the cage ceiling, or sleeping, signals were not generated (Fig. 1D). For drinking behavior to generate electrical signals, the metal lid and the metal sipper tube must form metal contact (Fig. 1B). Note that the signals were generated only by direct physical contact between animal and metal materials, but not by the induction. This was confirmed by putting the thin plastic barrier between the cage between metal lid and cage body (data not shown). Taken together, the recorded signals reflect HKA of an animal which can serve as a marker for circadian rhythm.
The currently available WRA-based circadian rhythm systems have cost issues and intrinsic limitations due to the dependence of a running-wheel. The cost issues arise from the need of purchasing additional sensors and recording equipment. Intrinsic limitation of WRA system is that wheel-running behavior itself alters the animal's activity patterns in some animals. To overcome these issues caused by wheel dependency, we have designed a HKA-based comprehensive circadian rhythm recording system with hardware and software (Fig. 2).
Circadian rhythm behavior experiments require relatively long period of recording time ranging from one month to a year. Thus, the stability of recording environment, temporal precision and convenience of management are important. In our HKA system, since the only required recording device is an A/D converter and itself serves as a sensor, signal quality and data acquisition stability were determined by A/D converter. For our HKA configuration, we utilized a standard A/D converter which was originally designed for electrophysiology (see details in Materials and Methods). This A/D converter, Digidata 1440a, can digitize 16 independent channels simultaneously, at sampling rates up to 250 kHz, and supported by AxoScope 10 software. AxoScope is a freely available, easy-to-use, full-featured data acquisition program for Windows. With these resources, we developed a HKA-based hardware configuration (Fig. 2A). We recorded signals in gap-free mode at the sampling frequency at 20 Hz, because this particular sampling frequency reliably detected “touching activities” while maintaining the reasonable size of the recorded files. Files were saved at 24 hour interval as “.abf (axon binary format)” file which is the default file format of Axoscope. For 16 channel recordings with duration of 24 hours, the size of files for 16 channels was 52.7 MB, indicating that at least 1.6 GB storage space is required for 30-day experiment.
To extract animal's touch activities from the raw electrical signals and visualize animal's circadian rhythmic activity patterns, we developed MATLAB-based user-friendly software, Circa Analysis (Fig. 2B). We also made a demonstration video to show step-by-step procedure of how to use the program (Supplementary Video 1). With “Extract” function, Circa Analysis converts raw signals to scalable “touch activities”. For example, when an animal touches the metal lid three times (for 2 s, 25 s and 15 s) during 0.1 hour (=360 s) of time bin, the total duration of touch activities is 42 s per bin. With “Display” function, Circa Analysis displays converted scalable “touch activities” to draw actogram by aligning the daily activities in a series (Fig. 2B). Taken together, with a unique combination of hardware and software based on HKA, we have developed a comprehensive circadian rhythm system.
To convert raw electrical signals into scalable touch activity, we applied various signal processing algorithms to the raw data. We recorded HKA with 20 Hz sampling frequency, and our raw data was masked by low frequency baseline noise (Fig. 3A, H, I) and this noise was completely removed when we applied Butterworth 1 Hz cut-off High Pass Filter (Fig. 3B and Formula 1). Although junction potential due to touch activity should be a positive value, our filtered data showed both positive and negative values. This is probably caused by HPF artifacts. To get rid of negative values, we have absolutized the signals (Fig. 3C and Formula 2). Next, we reduced the data size by averaging the number of samples at lower sampling frequency (Fig. 3D and Formula 3). For example, since our original sampling frequency was 20 Hz, averaging values of 20 consecutive samples made one data point per one second. This process simplified the handling of data. Next, to automatically discriminate the real touching signal from the baseline, we have applied a threshold based on Rosin's method [25] (Fig. 3G and Formula 4). Rosin's method is known as an effective auto-thresholding algorithm for unimodal histograms. It turned out that the amplitude frequency histogram of the reduced data showed a distribution of unimodal histogram, and therefore, optimal for applying Rosin's method. To explain briefly, the algorithm first draws a straight line from the peak to the end of the unimodal histogram. Second, the algorithm draws an orthogonal line from the peak-to-end line to each bin value of the histogram, until it finds the bin with a maximal distance from the peak-to-end line. When the distance is maximal, then Formula 4 becomes true. The threshold is determined as the amplitude value at this bin with maximal distance. Using this algorithm, we were able to search the optimal threshold automatically for each channel data. Using the threshold, we converted the reduced data into binary data, assigning the value of one of each data point if amplitude is greater than threshold, otherwise the value was set as zero (Fig. 3E and Formula 5). To quantify the duration of touch activities, we summated touch durations per bin interval (Fig. 3F and Formula6). In our analysis, we set the bin interval to 0.1 hour (=360 s). Thus the value of y axis represents total duration of touch activities per bin interval of 360 s. Taken together, with these six signal processing steps, we were able to automate the process of extracting numerical values for touch activities.
According to Nyquist-Shannon theorem, one's sampling rate should be twice greater than the target signal's bandwidth frequency (fS≥2fB). Therefore, to set the optimal range of recording sampling rate, it is crucial to understand the property of signals that we are handling. In our HKA system, we are recording the animal's touching activity. Since HKA signals are generated by animal's touching or licking of metal materials, the expected HKA signal bandwidth would be determined by maximal frequency of those behavior. Previous studies have reported that animal's licking frequency range is 4~7.5 Hz [24,26] and touching frequency range is 0.1~1 Hz [27]. Therefore, we expected the bandwidth of HKA signals to be less than 10 Hz and 20 Hz of sampling rate would be sufficient for detecting the HKA signals.
With 20 Hz sampling rate, the HKA signals were accurately detected along with animal's metal contact. However, as shown in the Fig. 3A and 3I, our signals of interest were contaminated with unidentified low frequency noise (<1 Hz). We found that the source of this noise was 60 Hz powerline interference signal which was aliased down to low frequency signal. The reason why 60 Hz signals were aliased down to under-1 Hz-signals was because our 20 Hz sampling rate is subnyquist sampling rate for 60 Hz signals. Since theoretically calculated aliasing frequency of 60 Hz signal sampled with 20 Hz is 0 Hz, we should not observe any baseline noise in recordings. However, 60 Hz powerline in real life has been shown to vary as much as ±1 Hz [28], and this drifts in 60 Hz frequency seem to be the major cause for our HKA system's baseline noise generation.
To confirm this idea, we simulated recording of drifting 60±1 Hz powerline interference with 20 Hz sampling rate (Fig. 3H). When 60 Hz signal was sampled with 20 Hz signal, the aliasing frequency was 0 Hz. Along with increase or decrease in 60 Hz powerline frequency, aliasing frequency increased exactly same with the step size. As long as drifts of 60 Hz signals are restricted to a range of ±1 Hz, our baseline noise frequency is less than 1 Hz. Based on simulation result, it is possible to remove low frequency baseline noise by 1 Hz HPF.
Before applying 1 Hz cutoff HPF to remove baseline noise, we had to confirm that whether our HKA signals would retain after filtering. By investigating the frequency domain and comparing “
Although our post-processing method provide much simpler way to setting up HKA system, note that adding a low-pass filter with cutoff frequency less than 60 Hz before ADC could have removed powerline interference at the beginning.
To verify that our system is working properly, we applied various protocols for circadian rhythm behaviors. First, we performed a phase advance experiment by applying 6 hour light pulse stimulation at Zeitgeber time (ZT) 15 of 11th day of constant dark period (Fig. 4A). Our HKA recording system successfully recorded mouse's phase advance (CT 3.1 h) of circadian rhythm without changing taus (before:23.5, after:23.69). Similarly, phase delay experiment by light pulse stimulation at 10th day of constant dark period showed mouse's phase delay (CT 3.5 h) of circadian rhythm without changing taus (before:23.7, after:23.8) (Fig. 4B). Next, we tested a jet-lag protocol (Fig. 4C) and showed actogram and Gaussian-filtered activity center plot. During jet-lag delay and advance period, animal spent 5 days and 4 days each to adapt. Lastly, seasonal variation protocols with light hour:dark hour ratio of 16:8 (Fig. 4D) and light hour:dark hour ratio of 8:16 (Fig. 4E). All of these experiments were stably and reliably recorded with HKA system. In addition, we confirmed that at least it is possible to record simultaneously from 16 mice over 150 consecutive days (data not shown). This indicates that HKA system can provide highly stable recording system for circadian rhythm behaviors.
To assess the cost-effectiveness of HKA system, we compared required components for WRA system and HKA system. As shown in Table 1, one can save the cost required for physical parameter counter (running-wheel) and transducer sensor (wheel-counter) with HKA configuration. Moreover, since HKA configuration utilizes distributional types for housing cage and A/D converter which are readily available in markets, HKA system can also benefit from not using company-tailored types which are optimized only for specific WRA system. Lastly, since we have developed a software which is optimized for this system, we were able to save additional cost for analysis software which is generally provided by company with a certain price. Overall, due to its simplicity of hardware composition, HKA system can be a highly cost effective system for recording circadian rhythm behavior.
DISCUSSION
For several decades, WRA configuration has been the most widely used system for circadian rhythm behavior research. In terms of the equipment price, however, it has been a financial barrier for some researchers. In this study, we present a new circadian rhythm behavior recording system that is easier to operate and more cost-effective than the existing WRA configurations (Fig. 1, Table 1). We have shown the principles of HKA recording system and developed a user-friendly software, Circa Analysis (Fig. 2). Our software provides automatic conversion of electrical signals into scalable touch activities in 6 signal processing steps (Fig. 3). In combination with various light-dark protocols, we have confirmed that the HKA recording system is capable of drawing actograms (Fig. 4) which are similar to those of currently available WRA configurations.
One of the great feature of HKA system is that there is no requirement of specific sensor. Generally, acquisition of data in most systems is mediated by following steps: 1) physical parameter detection (ex. wheel-running), 2) transducer sensor (ex. wheel counter), 3) A/D converter, 4) computer. However, in HKA system, simple A/D converter directly detects animal's natural behavior without any additional devices. The simplicity of data acquisition process is where HKA system has huge benefits over other recording systems that uses specific sensors [16,17,18,19,20,21,22,23].
As a result, this simple hardware composition in HKA configuration brings forth appreciable cost benefits. Since A/D converter not only serves as analogue to digital converter, but also as transducer sensor in HKA system, the complete HKA system can be established at a very low cost which is A/D converter. In this system, the estimated cost for recording circadian rhythm was less than 150$ per animal. If an A/D converter is already available, HKA configuration can be implemented at virtually no cost. Although our current HKA system utilizes a specific model of A/D converter and a recording software provided by a specific company (Molecular device), this can be overcome by replacing the A/D converter with one that can write “.abf” format files. This will allow the A/D converter to be compatible with our software, Circa Analysis. Therefore, future direction of our HKA system is to find a cost-effective A/D converter and develop complementary recording software for software compatibility with Circa Analysis.
HKA recording system is versatile and flexible. One of the great benefits for experimenters is that the maintenance of HKA system is much more convenient compared to WRA configurations because there are no accessories to maintain. Moreover, animals do not require additional habituation period for running-wheel housing cage. Since our system is recording natural house-keeping behaviors such as eating, drinking, and grabbing, there is virtually no possibility of changing animal's biological features caused by strenuous activities such as wheel-running [10,11,12,13,14,15]. Furthermore, HKA configuration is functionally expandable. One can record each animal's grabbing, drinking, and eating behavior separately by isolating the metal lid, metal food container, and metal sipper of water bottle and connecting to separate A/D converter channel to each metal part. Considering the fact that multifunctional metabolic cages cost tens of thousands of U.S. dollars per cage, HKA configuration system could be a great replacement for cost-effective multi-functional HKA recording system.
One of the disadvantages of our HKA configuration is that it is difficult to use any kind of wire-connected head mounting devices, such as fiber optic cables for optogenetics, wires for single unit-recordings and EEG recordings, and plastic tubings for microdialysis. However, this disadvantage applies equally to the WRA configurations. Using wireless devices could be a simple solution for this problem.
In conclusion, we propose our novel HKA-based circadian rhythm recording system as a low cost solution for circadian rhythm behavior experiment. Our HKA system reliably records circadian rhythm activities for a long duration without affecting animal's intrinsic properties.
SUPPLEMENTARY MATERIAL
Supplementary Video 1
Figures




Tables
| Categories | Object | WRA configuration | HKA configuration |
|---|---|---|---|
| 1) Animal housing | Housing cage | Running-wheel mountable tailored type | Distributional Type with grid metal floor 75.5$ / animalA) |
| 2) Physical parameter counting | Locomotion detector | Running-wheel | Not required |
| 3) Transducer sensor | Locomotion detector | Wheel-counter | Not required |
| Drinking detector (Optional) | Lick-sensor | Not requiredB) | |
| Feeding detector (Optional) | Feeding-monitor | Not requiredB) | |
| 4) Data converting | A/D converter | Tailored type of A/D converter compatible with wheel-counter | Any distributionalC) |
| 1025$ / 16 animalA) | |||
| 5) Data processing | Software | Company provided | No cost (Shareware) |
Comparison of essential components between WRA system and HKA system shows that WRA system requires running-wheel as a “2) Physical parameter counting” and wheel-counter as a “3) Transducer sensor”, whereas HKA system do not require any of those. In terms of “1) Animal housing” and “4) Data converting”, HKA configuration utilizes distributional types which are readily available, whereas WRA system requires tailored types which are optimized only for WRA system. For HKA system, we provide Circa Analyzer program for “5) Data processing” as shareware, whereas WRA system software is generally provided by company with a certain price.
A)The real cost in our HKA system. Total cost for 16 channel recording was 2225.0$ (139$/animal); B)It is also possible to measure feeding and licking exclusively by separating metal sipper, food container and metal cage ceil; C)Note that it is possible to measure electrical signals for natural activities, but our current software is only compatible for specific models of A/D converters provided by specific company (Molecular Devices). This standard A/D converter (Digidata 1440A) we used here is secondhand product, and this model is not available anymore at official distributors since new versions are released.
References
- Ko CH, Takahashi JS. Molecular components of the mammalian circadian clock. Hum Mol Genet 2006;15:R271-R277.
- Partch CL, Green CB, Takahashi JS. Molecular architecture of the mammalian circadian clock. Trends Cell Biol 2014;24:90-99.
- Takahashi JS. Transcriptional architecture of the mammalian circadian clock. Nat Rev Genet 2017;18:164-179.
- Panda S. Circadian physiology of metabolism. Science 2016;354:1008-1015.
- Bass J, Lazar MA. Circadian time signatures of fitness and disease. Science 2016;354:994-999.
- Man K, Loudon A, Chawla A. Immunity around the clock. Science 2016;354:999-1003.
- Musiek ES, Holtzman DM. Mechanisms linking circadian clocks, sleep, and neurodegeneration. Science 2016;354:1004-1008.
- Stephan FK, Zucker I. Circadian rhythms in drinking behavior and locomotor activity of rats are eliminated by hypothalamic lesions. Proc Natl Acad Sci U S A 1972;69:1583-1586.
- Jud C, Schmutz I, Hampp G, Oster H, Albrecht U. A guideline for analyzing circadian wheel-running behavior in rodents under different lighting conditions. Biol Proced Online 2005;7:101-116.
- Novak CM, Burghardt PR, Levine JA. The use of a running wheel to measure activity in rodents: relationship to energy balance, general activity, and reward. Neurosci Biobehav Rev 2012;36:1001-1014.
- Tachinardi P, Toien O, Valentinuzzi VS, Buck CL, Oda GA. Nocturnal to diurnal switches with spontaneous suppression of wheel-running behavior in a subterranean rodent. PLoS One 2015;10:e0140500.
- Blanchong JA, Mcelhinny TL, Mahoney MM, Smale L. Nocturnal and diurnal rhythms in the unstriped Nile rat, Arvicanthis niloticus. J Biol Rhythms 1999;14:364-377.
- Kas MJ, Edgar DM. A nonphotic stimulus inverts the diurnal-nocturnal phase preference in Octodon degus. J Neurosci 1999;19:328-333.
- Katona C, Smale L. Wheel-running rhythms in Arvicanthis niloticus. Physiol Behav 1997;61:365-372.
- Redlin U, Mrosovsky N. Nocturnal activity in a diurnal rodent (Arvicanthis niloticus): the importance of masking. J Biol Rhythms 2004;19:58-67.
- Bains RS, Wells S, Sillito RR, Armstrong JD, Cater HL, Banks G, Nolan PM. Assessing mouse behaviour throughout the light/dark cycle using automated in-cage analysis tools. J Neurosci Methods 2017
- Quinn LP, Stean TO, Trail B, Duxon MS, Stratton SC, Billinton A, Upton N. LABORAS: initial pharmacological validation of a system allowing continuous monitoring of laboratory rodent behaviour. J Neurosci Methods 2003;130:83-92.
- Goulding EH, Schenk AK, Juneja P, Mackay AW, Wade JM, Tecott LH. A robust automated system elucidates mouse home cage behavioral structure. Proc Natl Acad Sci U S A 2008;105:20575-20582.
- Steele AD, Jackson WS, King OD, Lindquist S. The power of automated high-resolution behavior analysis revealed by its application to mouse models of Huntington's and prion diseases. Proc Natl Acad Sci U S A 2007;104:1983-1988.
- Roughan JV, Wright-Williams SL, Flecknell PA. Automated analysis of postoperative behaviour: assessment of HomeCageScan as a novel method to rapidly identify pain and analgesic effects in mice. Lab Anim 2009;43:17-26.
- Mcshane BB, Galante RJ, Biber M, Jensen ST, Wyner AJ, Pack AI. Assessing REM sleep in mice using video data. Sleep 2012;35:433-442.
- Bains RS, Cater HL, Sillito RR, Chartsias A, Sneddon D, Concas D, Keskivali-Bond P, Lukins TC, Wells S, Acevedo Arozena A, Nolan PM, Armstrong JD. Analysis of individual mouse activity in group housed animals of different inbred strains using a novel automated home cage analysis system. Front Behav Neurosci 2016;10:106.
- Yaghouby F, Donohue KD, O'hara BF, Sunderam S. Noninvasive dissection of mouse sleep using a piezoelectric motion sensor. J Neurosci Methods 2016;259:90-100.
- Hayar A, Bryant JL, Boughter JD, Heck DH. A low-cost solution to measure mouse licking in an electrophysiological setup with a standard analog-to-digital converter. J Neurosci Methods 2006;153:203-207.
- Rosin PL. Unimodal thresholding. Pattern Recognit 2001;34:2083-2096.
- Weijnen JA. Licking behavior in the rat: measurement and situational control of licking frequency. Neurosci Biobehav Rev 1998;22:751-760.
- He J, Wei J, Rizak JD, Chen Y, Wang J, Hu X, Ma Y. An odor detection system based on automatically trained mice by relative go no-go olfactory operant conditioning. Sci Rep 2015;5:10019.
- Ramos R, Mànuel-Làzaro A, Del Río J, Olivar G. FPGA-based implementation of an adaptive canceller for 50/60-Hz interference in electrocardiography. IEEE Trans Instrum Meas 2007;56:2633-2640.



